Lupin Healthcare are committed to supporting the NHS in its goal of becoming net zero by 20401
Carbon Neutral Explained
Through carbon offsetting, Lupin Healthcare’s pMDIs portfolio has been certified as carbon neutral by Carbon Footprint Ltd.2-5
Every inhaler, including Dry Powder Inhalers (DPIs) has a carbon footprint associated with its production, and use6

Lupin Healthcare commissioned Carbon Footprint Ltd to complete Life Cycle Assessments (LCA) to understand the greenhouse gas (GHG) emissions associated with its pMDI portfolio.
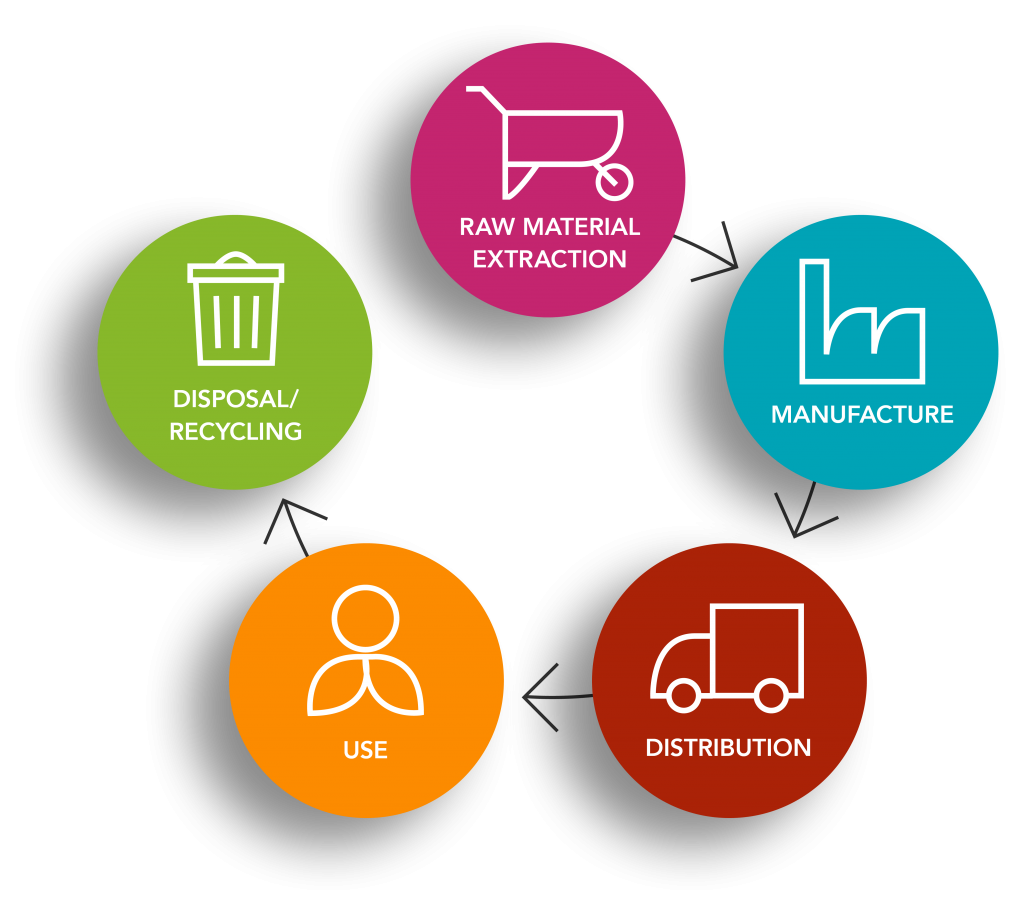
This assessment follows the principles of the Greenhouse Gas Protocol and ISO 14067:2018, and focus on the embodied GHG emissions of raw materials, the transport of these materials, the manufacture/processing, distribution, use and disposal of
Luforbec and Beclu pMDIs.2,3
By understanding the GHG emissions associated with the various stages of Luforbec’s life cycle, we can take steps to reduce these.
We offset the carbon footprint of all Lupin Healthcare pMDIs sold in the UK through supporting global sustainability and environmental projects. Through this process Luforbec and Beclu pMDIs have been certified as carbon neutral products by Carbon Footprint Ltd.2-5
For more information on the Carbon Footprint Standard requirements and methodology click here.
References: 1. NHS Delivering Net Zero Health Service https://www.england.nhs.uk/greenernhs/a-net-zero-nhs/ Accessed April 2023. 2. Carbon Footprint Limited, Luforbec Carbon Assessment Report 2022. Data on File. 3. Carbon Footprint Limited, Beclu Carbon Assessment Report 2023. Data on File. 4. Certifications of carbon neutrality for Luforbec 100/6 & 200/6 pMDI. 5. Certifications of carbon neutrality for Beclu 100 & 200 pMDI. 6. MIMS: Inhaler Carbon Emissions. https://www.mims.co.uk/inhaler-carbon-emissions/respiratory-system/article/1739635. Accessed: April 2023.
Contact Us
If you would like to get in touch, please contact us by the following
email: UKRespiratory@lupin.com or call: 01565 751378
This website uses essential cookies to ensure the adequate functioning of the website. More information about these cookies is available in the cookie section of our privacy policy. Click here to view our Privacy Policy.
UK-LUP-2208-00003 DOP: September 2022
UK-LUP-2208-00003 DOP: September 2022
Privacy & Cookies Policy
Privacy Overview
This website uses cookies to improve your experience while you navigate through the website. Out of these cookies, the cookies that are categorized as necessary are stored on your browser as they are essential for the working of basic functionalities of the website. We also use third-party cookies that help us analyze and understand how you use this website. These cookies will be stored in your browser only with your consent. You also have the option to opt-out of these cookies. But opting out of some of these cookies may have an effect on your browsing experience.
Necessary cookies are absolutely essential for the website to function properly. This category only includes cookies that ensures basic functionalities and security features of the website. These cookies do not store any personal information.
Any cookies that may not be particularly necessary for the website to function and is used specifically to collect user personal data via analytics, ads, other embedded contents are termed as non-necessary cookies. It is mandatory to procure user consent prior to running these cookies on your website.